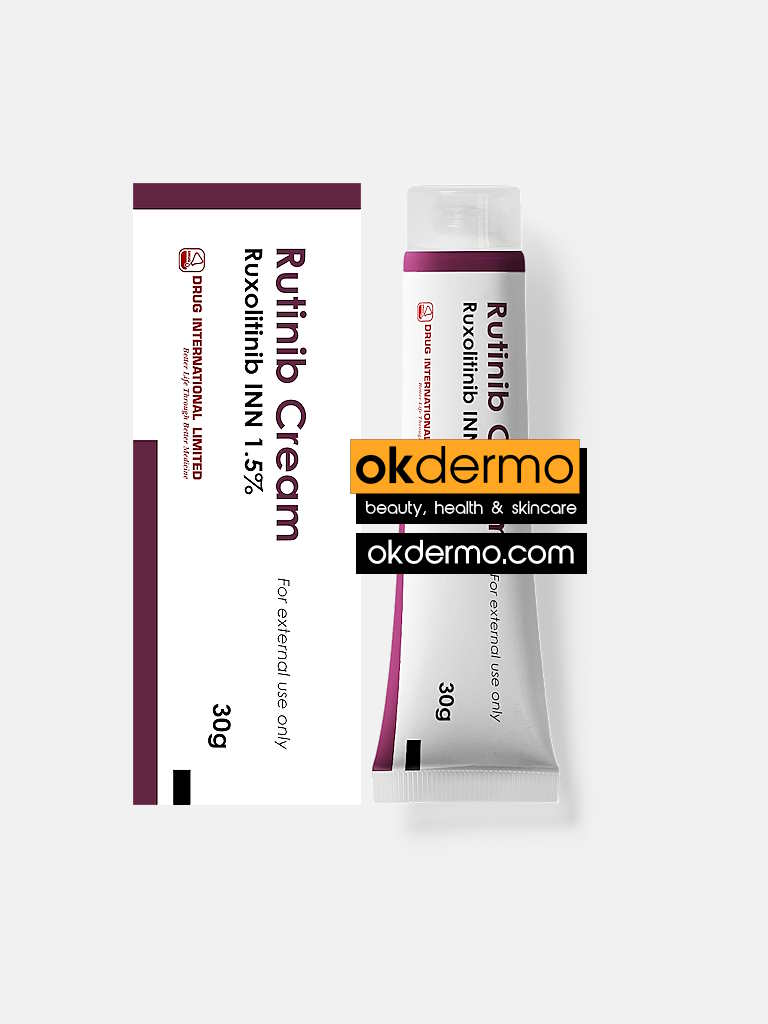
Description
What is Rutinib® Ruxolitinib Cream?
Welcome to our online pharmacy, your trusted destination for innovative skincare solutions. Uncover the transformative potential of Ruxolitinib Cream, marketed as Opzelura, a versatile remedy catering to vitiligo and more. Purchase Ruxolitinib Cream online and embark on a journey towards improved skin health. Secure your order now for rapid delivery.
Ruxolitinib Cream, in the form of Opzelura, brings new hope for vitiligo patients seeking effective relief. By targeting the underlying immune responses that trigger depigmentation, Ruxolitinib Cream assists in halting the progression of vitiligo and promoting repigmentation. Experience the potential of Opzelura to restore skin’s natural harmony and reduce the impact of this skin condition.
Beyond vitiligo, Ruxolitinib Cream has shown promise in addressing a range of inflammatory skin conditions. From psoriasis and eczema to atopic dermatitis, Ruxolitinib Cream’s targeted approach in inhibiting specific pathways of inflammation offers a ray of hope for patients experiencing discomfort and compromised skin health.
Opting for Ruxolitinib Cream, or Opzelura, signifies a commitment to comprehensive skin care. With its demonstrated efficacy and recognition as an innovative solution, Ruxolitinib Cream stands as a symbol of modern dermatology. By incorporating Ruxolitinib Cream into your skincare regimen, you’re taking a proactive step towards nurturing healthier, more vibrant skin.
Experience convenience and effective skincare when you buy Ruxolitinib Cream online from our user-friendly platform. Our secure payment gateway ensures a seamless transaction, providing peace of mind throughout your shopping journey. Don’t wait – secure your supply of Ruxolitinib Cream now and embark on a path towards enhanced skin well-being.
Elevate your skincare routine with Ruxolitinib Cream, known as Opzelura. Consult your healthcare provider, explore the benefits beyond vitiligo, and buy Ruxolitinib Cream online now. Our online pharmacy is dedicated to delivering transformative dermatological solutions directly to your doorstep.
Buy Ruxolitinib Cream Online: Unveil Transformative Skin Care with Opzelura. Order Now for Rapid Delivery!
FDA approved Ruxolitinib as an Opzelura Vitiligo Treatment
The Food and Drug Administration (FDA) has authorized Ruxolitinib (Opzelura), as the first at-home treatment for non-segmental vitiligo, an autoimmune condition that causes spots and patches of paler skin. Previously, it was used to treat atopic dermatitis, myelofibrosis, essential thrombocythemia, and polycythemia vera. It functions by lowering an individual’s enhanced immune response, gradually promoting the development of new, healthy skin cells, and ultimately reintroducing pigment to the afflicted area.
Using this topical cream twice daily can not only produce even skin tones but also boost patients’ self-esteem because vitiligo can be physically and psychologically upsetting. It is comparatively more efficacious and has a better safety profile than the oral forms of this medicine, although adverse effects such acne, redness, and itching at the application site, inflammation of the throat and nasal passages, headaches and fever have been observed, necessitating the need for observational studies and randomized controlled trials to demonstrate its efficacy and safety.
Nonsegmental Vitiligo causes and symptoms
Vitiligo is a chronic, autoimmune condition characterized by skin depigmentation and the formation of white patches on the face and body. The white patches can appear on any body part, such as hands, feet, arms, face, hair, scalp, eyebrows, lashes, beard, and mucus membrane inside the nose and mouth.
It is caused when the immune system attacks and destroys the pigment-producing cells, melanocytes.
The skin condition is classified into two major types, segmental and nonsegmental vitiligo. Nonsegmental vitiligo is more common than segmental vitiligo and generally appears symmetrically on both sides of the body.
Segmental vitiligo generally appears on only one side of the body and accounts for 5% to 16% of total vitiligo cases.
Vitiligo is estimated to impact between two and three million individuals worldwide, with nonsegmental vitiligo accounting for approximately 85% of the individuals.
Topical Ruxolitinib mechanism of action
Opzelura (Topical Ruxolitinib) is a typical JAK inhibitor that binds to JAK1 and JAK2 enzymes, involved in the signaling of a variety of cytokines and growth factors essential in hematopoiesis and immunological function. The binding results in the inhibition of IFN-γ-mediated JAK-signal transducers and activators of transcription (STAT) signaling, a key driver of depigmentation in vitiligo.
The inhibition is believed to reduce CD8+ T cell-mediated destruction of melanocytes, allowing a more stable environment for the return of melanocytes over time.
Clinical trials on Topical Ruxolitinib
The FDA approval of Opzelura was based on the positive outcomes of the pivotal Phase III TRuE-V clinical trial program, including TRuE-V1 and TRuE-V2 clinical studies.
The vehicle-controlled studies of Opzelura were double-blind, randomized, and of identical design to assess the cream’s safety compared to a non-medicated (vehicle) cream in more than 600 patients with nonsegmental vitiligo aged 12 and older.
In both studies, the patients were randomized at a 2:1 ratio to be treated with either Opzelura or vehicle cream twice daily for 24 weeks, followed by an additional 28 weeks.
The primary efficacy endpoint was the percentage of individuals who improved their vitiligo area scoring index (VASI) scores, which would indicate improvements in facial and total body repigmentation at week 24 compared to vehicle cream and in an open-label extension at week 52.
The facial (F)-VASI was used to evaluate lesions on the face while the total body (T)-VASI evaluated lesions on the body (including the face).
Results were consistent across both studies with approximately 30% of patients treated with Opzelura achieving at least 75% improvement in their F-VASI score (F-VASI75) at week 24, compared to approximately 8% and 13% of patients treated with the vehicle in TRuE-V1 and TRuE-V2, respectively.
Approximately 50% of patients treated with Opzelura achieved F-VASI75 at week 52.
The results demonstrated improvements in facial and total body repigmentation with Opzelura. The 52-week data also showed continued improvements in repigmentation with a longer duration of treatment with Opzelura.
The most common adverse reactions observed in patients treated with Opzelura were common colds, headaches, fevers, urinary tract infections, acne, itching, and redness at the application site.
How to use Rutinib® Ruxolitinib Cream (Opzelura Generic)
A thin layer of cream should be applied to the affected areas BID of up to 20% of body surface area; not to exceed >60 g/week. It should stop using when signs and symptoms (e.g., itch, rash, redness) of atopic dermatitis resolve. If signs and symptoms do not improve within 8 weeks, re-examine the patient. Or, as directed by the registered physician.


























Reviews
There are no reviews yet.