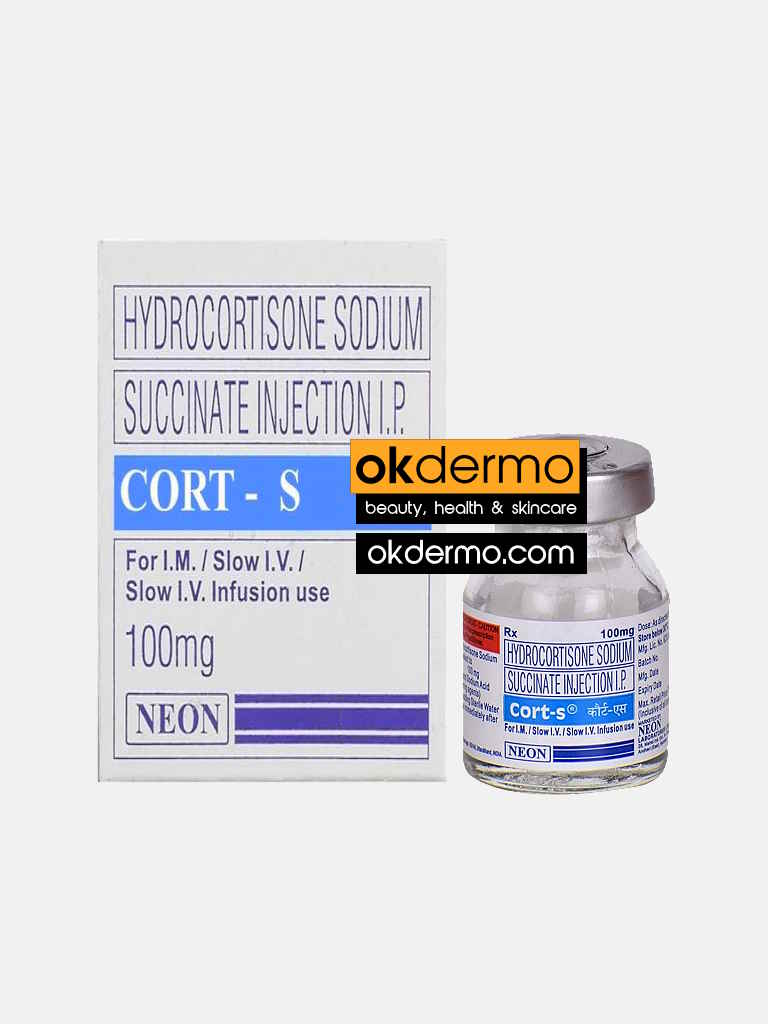
Description
What is Hydrocortisone Sodium 100mg Injection Cort-S®
Cort-S Injection (Hydrocortisone) is injected directly into a vein or into large muscles to treat a wide range of conditions including asthma, intestinal disorders, skin disorders, blood disorders, eye disorders, and thyroid disorders. It is also used to decrease the side effects of which can arise from taking other drugs. This treatment is part of the group of drugs known as cortisol, which have properties and effects similar to the corticosteroids or glucocorticoid hormones which reduce inflammation. Hydrocortisone Injection is used in the treatment of inflammatory skin conditions such as eczema, acne cysts and dermatitis, allergic skin reactions, and severe skin disorders.
Hydrocortisone Injection is known as a mild topical corticosteroid or topical steroid. Hydrocortisone Injection is used for treating and for the relief of symptoms caused by inflammatory skin conditions such as eczema and dermatitis and other allergic skin reactions. Always follow all instructions provided by your physician. Never self-medicate or alter your dosage by yourself. For soft tissue inflammation, 100mg to 200mg of Cort-S Injection (Hydrocortisone) is usually used. For other conditions please consult your physician. This treatment is usually administered by your physician, dermatologist.
Side effects of Hydrocortisone Sodium 100mg Injection Cort-S®
It is always important that your physician is informed of all side effects you notice during treatment with Cort-S Injection (Hydrocortisone / Cortisone). Notify your physician promptly if any persist, worsen, or become bothersome. Ensure your physician is aware if you are pregnant, trying to conceive a child, or breastfeeding, as this treatment may be unsafe.
Possible side effects may include headaches, vomiting, dizziness, mood swings, acne, facial redness, menstrual irregularities. The following side effects are more serious and require immediate medical attention: abnormal weakness, muscle pains, joint pains, vision changes or problems, any symptoms of an allergic reaction, such as swelling, skin rashes, hives, trouble breathing, and trouble swallowing.



























Elly Bee –
This drug saved my life
Aliney and Aaron –
Used it for adrenocortical insufficiency. I’m on 20mg daily split into 3 doses 10mg/ 5mg/5mg Keep my blood pressure stable and blood sugar levels But got long-term muscle weakness in my legs now So got to use crutches now Without this medication I wouldn’t be here to speak about this. So I’m really grateful I’m still here.