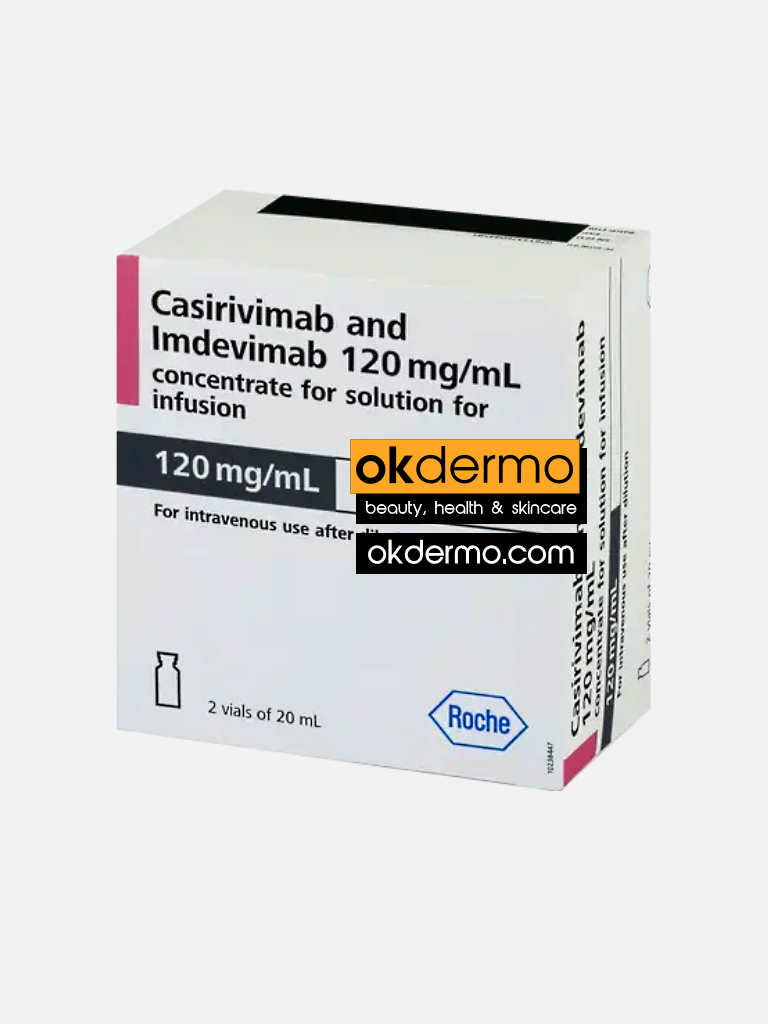
Description
What is Ronapreve® Casirivimab + Imdevimab Injection?
This medicine is made of casirivimab and imdevimab, two monoclonal antibodies. A monoclonal antibody is a type of protein that has been designed to recognize and attach to a specific structure (called an antigen). Casirivimab and imdevimab have been designed to attach to the spike protein of SARS-CoV-2 at two different sites. When the active substances attach to the spike protein, the virus is unable to enter the body’s cells.
Ronapreve 12mg injection is a combined injection that contains two antibodies belonging to monoclonal antibodies. Ronapreve injections are composed of Casirivimab and Imdevimab. Ronapreve injections are used in emergency management of mild to moderate symptoms associated with SARS-CoV-2 infections. Ronapreve injections contain two kinds of monoclonal antibodies known as Casirivimab and Imdevimab. This injection is only indicated for adults and children over 12years. Children over 12 years must have a minimum of 40kg in their body weight. The Ronapreve becomes more effective when given in the early days of diagnosis.
Ronapreve injections are administered intravenously or subcutaneously. The injection must only be administered by a skilled, trained professional. Ronapreve injections are not approved to use outside a hospital setup. This is because the direct injection of monoclonal antibodies can result in adverse reactions. Some of these adverse reactions need immediate medical treatments. So it must be administered in a hospital setup where emergency management can be obtained. A health professional must do close monitoring for up to one hour period to ensure the patient’s safety.
Monoclonal antibodies are prepared within a laboratory setup using cloning technology. White blood cells are used to prepare the monoclonal antibodies. So there is a higher chance of becoming allergic or hypersensitive to the Ronapreve injections. Never use this injection for self-injection purposes or never use it without a prescription. Your physician has to explore your history regarding previous health conditions and current situation before making any decision. Ronapreve injection comes as a clear solution, colorless of slightly pale yellow.
Coronavirus is composed of spiky proteins on its surface. Ronapreve can disturb the function of these proteins and prevent the entering of virus cells into human cells, So it helps the body get rid of the viruses and avoid infection with the SARS-CoV-2 virus.
The benefit of using Ronapreve injections
Ronapreve injections are aimed at treating mild to moderate SARS-CoV-2 infections. The monoclonal antibodies within the Ronapreve injections help in suppressing the infection. Therefore the use of Ronapreve injections at the initial stages can prevent severe respiratory symptoms. Severe respiratory depression may be needed to treat with supplemental oxygen. Ronapreve injections can control the turning of SARS-CoV-2 infections into harsh conditions where supplemental oxygen is needed for survival. When you feel that you have been exposed to the SARS-CoV-2 infection, go and discuss with the physician to confirm whether you need the injection or not.
Side effects of Ronapreve® Casirivimab + Imdevimab Injection?
Most of the simple side effects do not require remarkable medical advice. But remember to consult a physician if the side effects worsen with time.
- Injection site inflammation
- Fever with raised body temperature
- Flushing sensation around face, neck, and trunk
- Chest tightening
- Nausea and vomiting
How to use Ronapreve injection?
Never administer the Ronapreve injection by yourself. Do it under professional supervision. The injection must be performed by a trained health care professional. The skilled professional will follow the correct guidelines in dosing and administration.
Precautions:
- There are no studies conducted to explore the effect of alcohol with Ronapreve injections. So please try to avoid alcohol consumption soon after the injection.
- The information related to pregnancy and breastfeeding is also unavailable. So discuss with the physician if you are a pregnant woman or a breastfeeding mother.
- If you miss the dose, never double the amount.
- Be cautious if you have any underlying medical conditions. Disclose all the information to the physician.
How is Ronapreve® Used?
Ronapreve is given as a single treatment by infusion (drip) into a vein or by injection under the skin. The recommended dose is 600 mg of casirivimab and 600 mg of imdevimab.
When used for treatment, it should be given within 7 days of the patient developing symptoms of SARS-CoV-2.
When used for prevention after contact with a person with SARS-CoV-2, Ronapreve should be given as soon as possible after contact occurred. Ronapreve may also be given to prevent SARS-CoV-2 when no contact has occurred. In these cases, following an initial dose of 600 mg casirivimab and 600 mg imdevimab, a dose of 300 mg of casirivimab and 300 mg of imdevimab may be given every four weeks until prevention is no longer required.
The medicine can only be obtained with a prescription and should be given in healthcare facilities where patients can be adequately monitored and managed in case they develop severe allergic reactions, including anaphylaxis. For more information about using Ronapreve, see the package leaflet or contact your healthcare provider.





















Reviews
There are no reviews yet.