Description
What is Crisanta® Tablets?
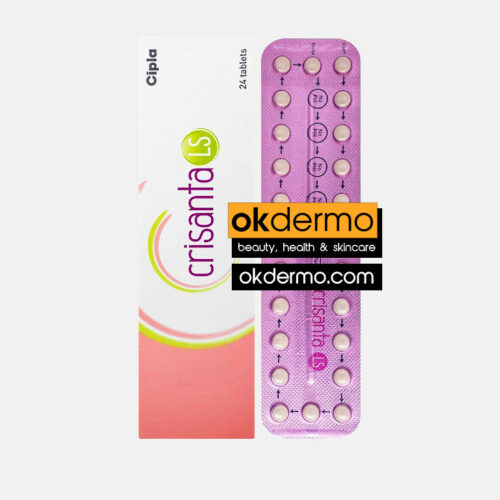
Crisanta® Tablets is a combined oral contraceptive pill. It works by preventing the release of an egg (ovulation) and affecting sperm movement in the womb to prevent its union with the egg. It also changes the lining of the womb and renders it unsuitable for pregnancy. Crisanta® birth control pill is manufactured by Cipla Ltd., India, and is offered to the consumer in a blister pack of 21 tablets. Each of the 21 pills of Crisanta® contains two active ingredients Drospirenone and Ethinyl Estradiol. Each film-coated Crisanta tablet contains 3 mg of Drospirenone and 0.03 mg of Ethinylestradiol.
Crisanta® contraceptive tablet is an example of such forefront innovation that offers the best in terms of efficacy, coupled with the least possible side effects. Combined oral contraceptives are highly effective when administered correctly without missing any pills, have a failure rate of approximately 1% per year (1 pregnancy per 100 women per year of use).
Besides contraceptive role, Crisanta® provides advantageous uses in varied other disorders of women’s health care such as treatment of moderate acne vulgaris in women who seek oral contraception.
This medicine also helps in the treatment of symptoms of the premenstrual dysphoric disorder (PMDD) in women who have chosen oral contraceptives as their method of birth control.
OKDERMO also has in stock Yaz Birth Control Pill which is another combination tablet used for the same indications.
How Does Crisanta® Contraceptive Pill Work?
Crisanta® lowers the risk of becoming pregnant primarily by suppressing ovulation. Other possible mechanisms can include cervical mucus changes that inhibit sperm penetration and endometrial changes that decrease the likelihood of implantation.
Crisanta® is a Combined Oral Contraceptive with ethinylestradiol and the progestogen, drospirenone. In therapeutic dosage, Drospirenone, a spironolactone analog, also possesses anti-androgenic and mild anti-mineralocorticoid properties. It has no estrogenic, glucocorticoid, and anti-glucocorticoid activity. This gives Drospirenone a pharmacological profile that closely resembles the natural hormone, progesterone.
Crisanta® Contraceptive Pill Dosage and Administration
In order to achieve maximum contraceptive effectiveness, the Crisanta® birth control pill must be taken as directed in the order on the blister pack. A patient should start taking Crisanta® on the first day (Day 1) of her menstrual period.
During the first cycle of Crisanta® use, the patient must be instructed to take one tablet daily, beginning on day 1 of her menstrual cycle (the first day of menstruation is day one). She should take one tablet of Crisanta® daily for 21 consecutive days, followed by 7 pill-free days. It is recommended that Crisanta® be taken at the same time each day, preferably after the evening meal or at bedtime.
If Crisanta® administration is started later than the first day of the menstrual cycle, it should not be considered as an effective contraceptive until after the first 7 consecutive days of administration of this medication. The patient should use a non-hormonal contraceptive as backup during the first 7 days. The possibility of ovulation and conception prior to initiation of medication should be considered.
The patient should begin her next and all subsequent 28-day regimens of Crisanta® on the same day of the week that she began her first regimen, following the same schedule. She should begin taking her tablets on the next day after the 7 pill-free days, regardless of whether or not a menstrual period has occurred or is still in progress. Any time a subsequent cycle of Crisanta® is started later than the day following 7 pill-free days, the patient should use another method of contraception until she has taken a tablet daily for 7 consecutive days.
When Switching from a Different Birth Control Pill. When switching from another birth control pill, Crisanta® tablets should be started on the same day that a new pack of the previous oral contraceptive would have been started.
When Switching from a Method other than a Birth Control Pill. When switching from a transdermal patch or vaginal ring, Crisanta® tablets should be started when the next application would have been due. When switching from an injection, Crisanta® tablets should be started when the next dose would have been due. When switching from an intrauterine contraceptive or an implant, Crisanta® tablets should be started on the day of removal.
Crisanta Crisanta® Birth Control Pill Missed Pill Instructions
The efficiency of the Crisanta® contraceptive pill relies on the motivation of the patient. Daily intake promotes adequate blood hormone levels ensuring safety against pregnancy.
Missed Pill. If the patient is less than 12 hours late in taking any tablet, contraceptive protection is not reduced. The woman should take the tablet as soon as she remembers and should take further tablets at the usual time.
If she is more than 12 hours late in taking any tablet, contraceptive protection may be reduced. The management of missed tablets can be guided by the following two basic rules:
Tablet-taking must never be stopped for more than 7 days. 7 days of uninterrupted tablet-taking are necessary in order to attain adequate suppression of the hypothalamic-pituitary-ovarian axis. Accordingly, the following advice can be given in daily practice:
Week 1: The user should take the last missed tablet as soon as she remembers, even if this means taking two tablets at the same time. She then continues to take tablets at her usual time. In addition, a barrier method such as a condom should be used for the next 7 days. If intercourse took place in the preceding 7 days, the possibility of a pregnancy should be considered. The more tablets are missed and the closer they are to the regular tablet-free interval, the higher the risk of pregnancy.
Week 2: The user should take the last missed tablet as soon as she remembers, even if this means taking two tablets at the same time. The patient then continues to take tablets at her usual time. Provided that the woman has taken her tablets correctly in the 7 days preceding the first missed tablet, there is no need to use extra contraceptive precautions. However, if she has missed more than 1 tablet, the woman must be advised to use extra precautions for 7 days.
Week 3: The risk of reduced reliability is imminent because of the forthcoming 7-day tablet-free interval. However, by adjusting the tablet-intake schedule, reduced contraceptive protection can still be prevented. By adhering to either of the following two options, there is, therefore, no need to use extra contraceptive precautions, provided that in the 7 days preceding the first missed tablet the woman has taken all tablets correctly. If this is not the case, she should follow the first of these two options and use extra precautions for the next 7 days as well.
The user must take the last missed tablet as soon as she remembers, even if this means taking two tablets at the same time. She then continues to take tablets at her usual time. The next blister pack must be started as soon as the current blister pack is finished, i.e., no gap should be left between packs. The patient is unlikely to have a withdrawal bleed until the end of the second pack, but she may experience spotting or breakthrough bleeding on tablet-taking days.
The patient may also be advised to discontinue tablet-taking from the current blister pack. She should then have a tablet-free interval of up to 7 days, including the days she missed tablets, and subsequently, continue with the next blister pack.
If the woman missed tablets and subsequently has no withdrawal bleed in the first normal tablet-free interval, the possibility of pregnancy must be considered.
Crisanta® Overdosage
There have been no reports of serious adverse effects from Crisanta® overdose, including ingestion by children. On the basis of general experience with Combined Oral Contraceptives, symptoms that may possibly occur in this case are nausea, vomiting, and, in young girls, slight vaginal bleeding. There are no antidotes and further treatment should be symptomatic.
Drospirenone (DRSP) is a spironolactone analog that has anti-mineralocorticoid properties. The serum concentration of potassium and sodium, and evidence of metabolic acidosis, have to be monitored in cases of overdose.
Side Effects of Crisanta® Tablets
Minor reactions are known to occur use of Crisanta® tablets and these are not serious at all and usually disappear on their own or may need light treatment.
The most frequent Crisanta® side effects with an occurrence rate of 2% or more, when used for contraception and acne treatment, are headache/migraine (6.7%), menstrual irregularities (4.7%), nausea/vomiting (4.2%), breast pain/tenderness (4.0%) and mood changes (2.2%). The other possible side effects of Crisanta® tablets are given below:
- Amenorrhoea: Post pill amenorrhoea or a complete absence of menses/ withdrawal bleed is quite common in habitual and regular pill users over longer periods of time. Nevertheless, if pill intake is regular pregnancy is not a consideration.
- Breakthrough Bleeding: Hormonal levels in the blood must be maintained above a certain threshold. Fluctuating levels weaken the endometrial architecture and easy shedding results manifest as spotting to light bleeds during pill intake days.
- Nausea, lack of appetite, abdominal cramps, bloating, and rarely vomiting.
- White vaginal discharge or Leucorrhoea, Yeast/ fungal infections.
- Headaches, hair loss, Insomnia, Nervousness, Mood swings, changes in libido.
- Allergy, Rash, fever, fatigue, anorexia.
- Darkening of facial skin, breast discomfort, and congestion.
Crisanta® Tablets Contraindications
Combined oral contraceptives (COCs) like Crisanta® tablets should not be used in the presence of the following situations listed below:
- Hypersensitivity to Drospirenone or Ethinyl Estradiol or to any of the inactive ingredients contained in Crisanta® tablets
- The presence or a history of venous or arterial thrombotic/thromboembolic events (e.g. deep venous thrombosis, pulmonary embolism, myocardial infarction) or of a cerebrovascular accident.
- Presence or history of prodromi of a thrombosis (e.g. transient ischaemic attack, angina pectoris)
- The presence of a severe or multiple risk factor(s) for venous or arterial thrombosis may also constitute a contraindication.
- History of migraine with focal neurological symptoms
- Diabetes mellitus with vascular involvement
- Pancreatitis or a history thereof if associated with severe hypertriglyceridemia
- Presence or history of severe hepatic disease as long as liver function values have not returned to normal
- Presence or history of liver tumors (benign or malignant)
- Severe renal insufficiency or acute renal failure
- Known or suspected sex-steroid influenced malignancies (e.g. of the genital organs or the breasts)
- Undiagnosed vaginal bleeding, known or suspected pregnancy.
Should any of the conditions listed above appear for the first time during the use of this medicine, the product should be stopped immediately.
Interactions of Crisanta® with Other Drugs
- Important drugs that reduce the effectiveness of Crisanta® and other oral contraceptives include Anticonvulsants Carbamazepine (Tegretol), Phenytoin (Dilantin), Lamotrigine (Lamictal), Phenobarbitone (Gardenal).
- Antibiotics Rifampicin (used in tuberculosis), Ampicillin, Metronidazole (Vaginal infection, loose motions), and Nitrofurantoin.
- Hence it is vital to inform your treating doctor of all the medications you are taking prior to starting Crisanta® as a reduction in its efficacy from any of the above agents may lead to an unsuspected pregnancy.
Precautions while on Crisanta® Tablets
The following precautions must be noted when using Crisanta® tablets:
- Crisanta® does not offer any protection against sexually transmitted diseases or HIV and hence while on this medicine, barrier contraceptives must be used to guard against these.
- Crisanta® tablets will not harm any inadvertent pregnancy if it occurs while on it but must be stopped immediately if wanting to continue the pregnancy.
- Crisanta® affects lactation by reducing the amount of breast milk produced and alternative contraception methods must be used during breastfeeding.
Crisanta® Tablets During Pregnancy
There is little or no increased risk of birth defects in women who inadvertently use Combined oral contraceptives (COCs) like Crisanta tablets during early pregnancy.
Epidemiologic studies and meta-analyses have not found an increased risk of genital or non-genital birth defects (including cardiac anomalies and limb-reduction defects) following exposure to low-dose COCs prior to conception or during early pregnancy.
The administration of Combined oral contraceptives to induce withdrawal bleeding should not be used as a test for pregnancy. Combined oral contraceptives should not be used during pregnancy to treat threatened or habitual abortion.
Stop using this medication and inform your doctor immediately if you become pregnant during treatment.
Nursing Considerations
When possible, advise the nursing mother to use other forms of contraception until she has weaned her child. Estrogen-containing combined oral contraceptives can reduce milk production in breastfeeding mothers. This is normally less likely to occur once breastfeeding is well-established. However, it can happen at any time in some women. Small amounts of oral contraceptive steroids and/or metabolites are present in breast milk.
After oral administration of 3 mg Drospirenone (DRSP) /0.03 mg Ethinyl Estradiol (EE) tablets, about 0.02% of the DRSP dose was excreted into the breast milk of postpartum women within 24 hours. This results in a maximal daily dose of about 0.003 mg DRSP in an infant. Do not use this medication without telling your doctor if you are breastfeeding a baby.
Buy Cipla Crisanta® Contraceptive Tablets Online at cheap
You can buy Crisanta® Contraceptive tablets online from okdermo.com. Crisanta® birth control pills, manufactured by Cipla Ltd., India are supplied in a box of 21 tablets. The tablets containing 3 mg of Drospirenone and 0.03 mg of Ethinyl Estradiol cost only $23 per 21 Tablet Blister when you place an order with us.