Description
What is 3% / 5% Tranexamic Acid Gel Tranesma®
Tranesma® skin care gel is manufactured by reputable Akumentis Healthcare. The gel contains the main active component – Tranexamic acid 3 percent strength. Tranexamic acid slows melanin synthesis by inhibiting the plasminogen/plasmin pathway. Doing so blocks interactions between melanocytes and keratinocytes. Consequently, using either oral or topical tranexamic acid can decrease melasma-associated skin pigmentation and reverse any melasma-related dermal changes, including reduced redness and a decreased number of vessels. Tranexamic acid is successfully used for the treatment of melasma as a safer alternative to hydroquinone.
What is Tranexamic acid generally used for?
Listed in the World Health Organization’s list of Essential Medicines1, tranexamic acid was originally used to reduce blood loss in patients experiencing menorrhagia and during open-heart surgery. However, in 1979, physicians accidentally began noticing that patients who had been treated with tranexamic acid were seeing visible improvement in skin discoloration. More recently, tranexamic acid has been introduced as a topical skincare ingredient aimed to reduce the appearance of stubborn forms of discoloration. It is also used as an adjunct therapy to in-office procedures such as laser or intense pulsed light (IPL).
Benefits of Tranexamic acid 3% / 5% Gel
When shopping for products that target skin discoloration, it is important to look for well-studied ingredients backed by clinical results. Tranexamic acid is one such ingredient. Used and recommended by dermatologists, it works by addressing the appearance of discoloration at its onset. Studies have also demonstrated its ability to address hard-to-treat forms of discoloration including post-acne marks and stubborn brown patches. Tranexamic acid can be combined with other discoloration-fighting ingredients that work synergistically to create a multi-action approach targeting discoloration.
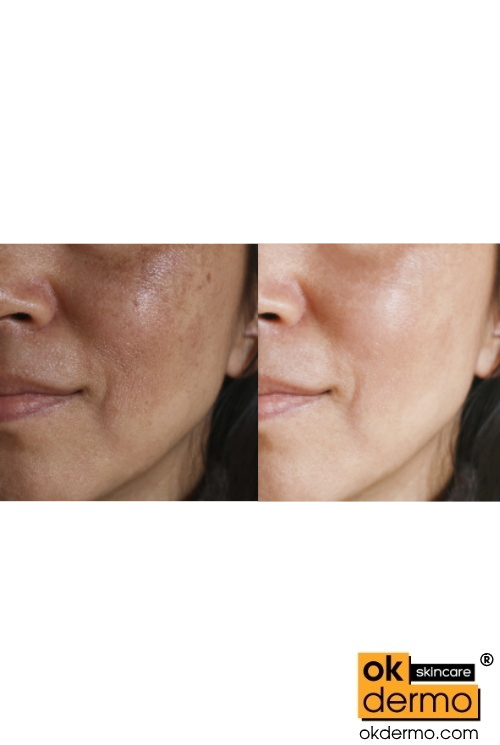
Tranesma by okdermo skin care is a daily-use gel formulated with 3% tranexamic acid. This clinically-proven formula improves the look of discoloration, brightens skin, and evens skin tone in as early as 2 weeks. For the most comprehensive regimen, it is always recommended to pair with a vitamin C serum and broad-spectrum sunscreen to protect and prevent UV-induced skin damage that can contribute to an uneven skin tone.
Mechanism of action of Tranexamic acid on Melasma
To understand how tranexamic acid helps brighten skin, it is important to understand why skin discoloration occurs in the first place. Everyday elements – including UV exposure, prescription drugs, and some professional treatments – can trigger the skin discoloration process by activating inflammatory mediators within the skin. This results in a chain reaction that causes the skin to produce melanin, the naturally-occurring pigment that gives skin its color. While the purpose of melanin is protective, when produced in excess, it can form deposits in the skin that manifest as dark patches, isolated spots, or an overall darker facial appearance.
Tranexamic acid is a potent ingredient that helps reduce the appearance of discoloration and improves the look of stubborn brown spots, resulting in an overall brighter appearance.
Effects of Topical Tranexamic Acid in Comparison with Hydroquinone in Treatment of Melasma
INTRODUCTION: Few studies have focused on therapeutic as well as side effects of tranexamic acid (TXA) as a topical drug compared to other topical drugs in treating melasma. The present study aimed to assess and compare the beneficial therapeutic effects and also side effects of local TXA in comparison with hydroquinone in treating women with melasma.
METHODS: This randomized double-blinded clinical trial was performed on 60 women who suffered from melasma and were referred to the skin disorders clinic at the Rasoul-e-Akram hospital in Tehran in 2015. The patients were then randomly assigned via computerized randomization to two groups: group A received TXA%5 (topically twice a day for 12 weeks in the location of the melasma) and group B (received hydroquinone 2% with the same treatment order). Prior to intervention and at 12 weeks after the intervention, the intensity and extension of melasma were assessed based on the Melasma Area and Severity Index (MASI) scoring method.
RESULTS: The mean MASI score in both treatment groups decreased considerably after the completion of treatment and was not significant between the two groups. No side effects were detected in group A, but 10% of those in group B complained of drug-related side effects including erythema and skin irritation. Regarding the level of patient satisfaction, the patients in group A had a significantly higher level of satisfaction level of 33.3% compared with 6.7% in group B. Multivariate linear regression modeling with the presence of age, history of systemic disorder, drug history, and family history of melasma demonstrated no difference in the mean MASI between the two groups.
CONCLUSION: Topical use of Tranexamic acid significantly reduced both melanin level and MASI score. Given its high efficiency and low drug side effects, this regimen results in high patient satisfaction compared with topical hydroquinone.
Reference: US National Library of Medicine National Institutes of Health
Tranexamic acid in melasma: Why and how?
Melasma is a chronic, acquired symmetrical pigmentary disorder characterized by gray-brown macules and patches affecting the photodistributed part of the face such as the bridge of the nose, cheek, upper lip, forehead, and mandible. It is seen mostly in women of reproductive age group and has an onset usually after pregnancy. However, in around 10% of the cases, males are also affected. Causative factors that have been implicated in aggravating or precipitating melasma include sex hormones, genetic susceptibility, ultraviolet (UV) light exposure, oral contraceptive pills, cosmetics, and phototoxic drugs. Epidemiologically, melasma is found in all geographical and ethnic groups but occurs more commonly in Asians and Middle Easterners.
Treatment is most unsatisfactory. Various modalities include hydroquinone (HQ), tretinoin, and corticosteroid triple combinations, nonsteroidal demelanizing creams, superficial chemical peels (glycolic acid, trichloroacetic acid, kojic acid, and lactic acid), lasers (Q-switched Nd:YAG laser, ruby laser, Alexandrite laser, Er:YAG laser, and Fraxel laser), and intense pulsed light (IPL).
Despite the plethora of treatment modalities, the results of these treatments in melasma are poor. Treatment is often incomplete and there is a high rate of recurrence. Tranexamic acid (TXA) has been recently recommended for the use in melasma. It can be given as an oral preparation, a topical preparation, or an intralesional agent. It is probably the first oral medication that has proven efficacy in the treatment of melasma. Other oral medications are Polypodium leucotomos which has an indirect effect, and glutathione which has yet unproven efficacy.
Tranexamic acid: TXA (trans-4-aminomethyl cyclohexane carboxylic acid) is an antifibrinolytic agent which blocks lysine-binding sites on plasminogen molecules. TXA is not a new molecule. It has been extensively used in the treatment of menorrhagia since the 70s. Dosages up to 2–4 g/day for 4–7 days have been used safely during cycles. TXA has also been used prophylactically in hereditary angioedema in the duration of 8–34 months.
For melasma, it has been used in low doses of 250 mg BD. Contraindications include defective color vision, coagulopathy, hypersensitivity, cardiovascular disease, stroke, and anticoagulant medications. Before starting TXA, it is important to get prothrombin time, activated partial thromboplastin time, and other blood clotting parameters. Common adverse effects include gastrointestinal side effects and reversible hypomenorrhea. Other reported adverse effects include deep vein thrombosis (DVT), myocardial infarction, and pulmonary embolism.
Use of Tranexamic Acid in Melasma: The use of Tranexamic acid in melasma was first reported by Nijo in 1979. It was an accidental discovery in a patient of melasma who was receiving TXA for chronic urticaria. Sun exposure of the skin leads to the synthesis of plasmin activator, which thereby increases plasmin activity in keratinocytes. This plasmin leads to release of arachidonic acid (AA) via phospholipase A2. Prostaglandin E2 synthesis promoted by AA stimulates melanogenesis. Increased plasmin elevates α-melanocyte-stimulating hormone and fibroblast growth factor which are both potent melanocyte stimulators. Plasmin also plays a role in angiogenesis by increasing the release of free vascular endothelial growth factor (VEGF).
Repeated UV damage leads to increased production of mast cell tryptase which weakens and damages the basement membrane, a condition seen in melasma. Contraceptive pills and pregnancy have also shown to increase serum plasminogen activator that can activate the melanogenesis process. Other dermal changes also seen in melasma include disruption of the basement membrane, increased blood vessels, and solar elastosis. A number of mast cells may also be increased in the lesional dermis. These dermal factors may be the cause behind the refractory nature of melasma. There are various reports and studies to authenticate the safety and usefulness of TXA in melasma. Some of these studies are mentioned here.
Conclusion: Oral Tranexamic acid at the dose of 250 mg twice daily is found to be a promising modality for the treatment of melasma. It is the only agent that acts by inhibiting UV light-induced melanocyte activation, hormone-induced melanocyte proliferation, VEGF-induced neovascularization, and mast cell-induced basement membrane damage. It can be used both as a standalone therapy and in conjunction with other modalities. It can improve the results achieved by lasers and IPL. Topical tranexamic acid is also effective and safe in combating melasma locally.
Considering effects of TXA at various levels of melanogenesis in melasma, it has a definite place in the treatment of this frustrating condition. Further studies are required to standardize the dose and duration of oral and topical Tranexamic acid in melasma. Large studies need to be done in various ethnic groups. In conclusion, Tranexamic acid has proven to be an essential tool in the armamentarium against melasma.
Reference: Dr. Davin Lim